Introduction
Indolent systemic mastocytosis (ISM), a clonal mast cell disease driven by the KIT D816V mutation in ~95% of cases, can cause lifelong debilitating cutaneous, gastrointestinal, and neurocognitive symptoms with some cases progressing to advanced systemic mastocytosis (AdvSM). While several studies have quantified variable rates of disease progression from ISM to AdvSM based on the absolute frequency of these events, these studies were limited by short median follow-up time considering the long clinical course of this condition sometimes in decades, thereby leading to underestimation of the cumulate rates of progression from ISM to AdvSM. We conducted a Markov analysis using population level data in the US to estimate the lifetime risk of progression for ISM patients.
Methods
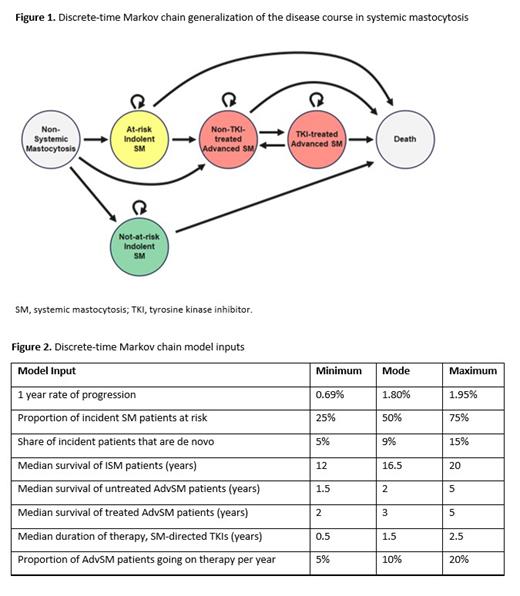
We modeled the total course of SM as a discrete-time Markov chain, enabling us to estimate cumulative rates of disease progression based on fixed-interval empirical observations from the literature and, where there is no literature available, expert opinion. The Markov model assumes that patients enter the model as at-risk ISM, not-at-risk ISM, or de novo AdvSM, based on the assumed relative incidence of each subtype. Not-at-risk patients will then move to death after a period of time, with an annualized risk of death from disease. At-risk patients with ISM will remain in that state, progress to AdvSM (which may be tyrosine kinase inhibitor treated or not) or die. Finally, those who progress to AdvSM will either remain in their state or die. See Figure 1 for details, where each model parameter is reflected as a line between states and modeled based on an annualized rate of state transition. See Figure 2 for a full list of model parameters. Two hundred trials were performed, in which the model parameters were statistically sampled from a range of likely values, based on a triangular distribution between minimum, mode, and maximum. For each trial, the disease course for 5000 patients with SM was then simulated from initial SM diagnosis until death.
Results
Based on the rates of disease incidence, progression, treatment, and survival documented within various studies, we estimate the cumulative risk (± standard deviation) of disease progression from ISM to AdvSM to be 18.0% ± 3.1%, based on a probabilistic sampling of the underlying assumptions.
Conclusions
This study suggests that the cumulative rate of disease progression among patients with ISM may be higher than previously recognized. In our earlier work, we showed a higher rate of disease progression in ISM patients with high symptom burden (Mukherjee S, et al. ASH 2022). These data highlight the importance of close monitoring for signs of progression and warrant further research to identify subsets of ISM patients who may have a higher underlying risk of progression.
Disclosures
Mukherjee:Novartis: Consultancy; Celgene (now BMS): Consultancy; BioPharm: Consultancy; EUSA: Honoraria; McGraw Hill Hematology Oncology Board Review: Honoraria; Bristol Myers Squibb: Honoraria; Celgene (now BMS): Honoraria; Aplastic Anemia and MDS International Foundation: Honoraria; EUSA: Other: Advisory Board; Genentech and AbbVie: Other: Advisory Board; Blueprint Medicines Corporation: Other: Advisory Board; Novartis: Other: Advisory Board; Bristol Myers Squibb: Other: Advisory Board; Celgene/Acceleron: Other: Advisory Board; Bristol Myers Squibb: Consultancy; Celgene (now BMS): Research Funding; Novartis: Research Funding; Jazz Pharmaceuticals: Research Funding. Cattie:Blueprint Medicines Corporation: Current Employment, Current equity holder in publicly-traded company. Shaheen:Blueprint Medicines Corporation: Current Employment, Current equity holder in publicly-traded company. Green:Blueprint Medicines Corporation: Current Employment, Current equity holder in publicly-traded company. Powell:Blueprint Medicines Corporation: Current Employment, Current equity holder in publicly-traded company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal